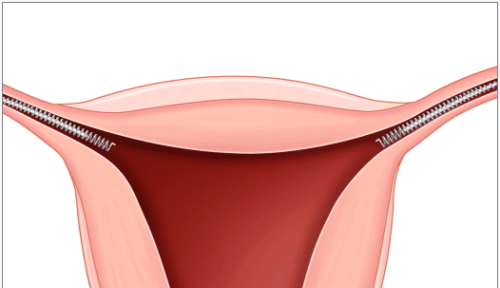
Essure is a hormone-free, permanent implant birth control for women. Essure is intended to be nonreversible and an alternative to tubal ligation (tubes tied). Essure is inserted into the fallopian tubes by a doctor. The procedure is typically performed in a doctor’s office and does not require general anesthesia.
While the device’s manufacturer claims Essure is safe and effective, thousands of women claim that Essure has caused them to experience serious and even life-threatening injuries.
If you had Essure inserted and you have suffered from one of the following complications, you may be entitled to compensation:
- Pain and bleeding
- Device migration
- Device fracture
- Organ perforation
- Complication requiring removal
- Allergic reactions
Essure Birth Control Class Action Lawyers
Walton Telken is currently offering free consultations for all our prospective Essure clients. We will let you know your options. If retained to represent you, we will assume all upfront financial risk – this means we cover all the expense of litigation and only charge a fee if we win your case. We file lawsuits in individual cases after review of your medical records and assessment of the facts in your case. We will work hard to obtain fair and adequate compensation for those that have suffered severe injuries related to their Essure implant.
Time limitations can bar claims, so if you believe Essure may have caused harm, please contact our firm as soon as possible. You can call us today, toll-free at 844-307-7349 for a free, no-risk consultation.
Related articles:
- https://www.revealnews.org/episodes/her-own-devices-is-a-contraceptive-implant-making-us-sick/
- http://www.washingtonpost.com/sf/style/2017/07/26/essure/?utm_term=.33e6cffc4889 (podcast embedded in article)
- https://www.cbsnews.com/news/bayer-essure-implant-sales-halted-outside-us/
Litigation Update:
On July 20, 2018, Bayer announced it will pull Essure off the market at the end of 2018. Essure has previously been removed from all other world markets due to safety concerns. This will prevent more women from experiencing the pain and loss due to this defective device. Bayer continued to put profits over women’s health, but Essure victims helped call attention to the dangers of this device. There are over 15,000 lawsuits filed across the country against Bayer related to the painful damages that Essure can cause including perforated organs, chronic pain, heavy bleeding, loss of hair, skin irritations and other painful side effects.
In response to Bayer’s announcement, the FDA released a statement. “For women who have received an Essure implant, the postmarket safety of Essure will continue to be a top priority for the FDA.” The FDA will continue to review all Essure adverse advent reports and monitor Bayer’s compliance with its responsibilities related to a postmarket study required by the FDA in 2016. https://www.fda.gov/